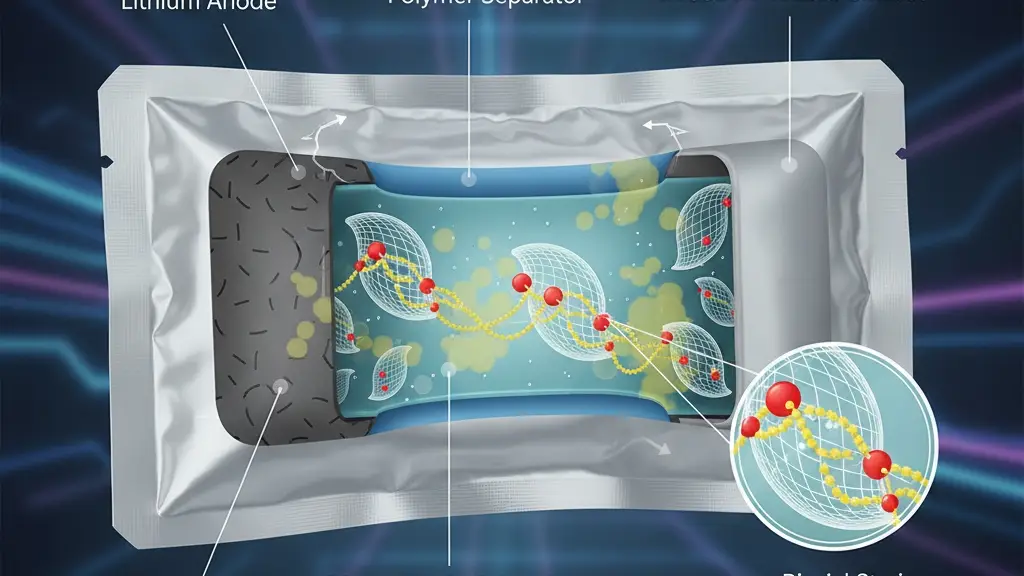
Lithium–sulfur (Li–S) batteries could store a lot of energy because sulfur carries an enormous theoretical capacity. But dissolved sulfur chains (“polysulfides”) wander around and gunk up the battery—like sugar drifting into your coffee’s foam—slowing reactions and wasting material.
A new Nature Communications study shows that if you curve and biaxially strain a 2D catalyst (a Mo-based sulfur/selenium compound), you expose buried molybdenum (Mo) atoms on the surface. Those Mo atoms act like sticky parking spots for sulfur species, so the reactions run faster and the wandering problem shrinks. In tests, the researchers built multilayer Li–S pouch cells with ~6.1 Ah total capacity and ~396 Wh/kg specific energy (counting everything in the pack), and they kept cycling well.
For Science Folks (but readable for everyone)
Why Li–S is tempting
- Theoretical specific capacity: 1675 mAh g⁻¹ (sulfur).
- Theoretical specific energy: ~2600 Wh kg⁻¹—an order of magnitude above many commercial Li-ion packs.
The core headache: the shuttle + slow kinetics
During discharge/charge, lithium polysulfides (Li₂Sx) form and dissolve. They diffuse (“shuttle”) to the anode and back, wasting sulfur and causing fade. Also, converting long-chain polysulfides into solid Li₂S can be kinetically sluggish—think traffic jam at a toll booth.
The new trick: high-dimensional (biaxial) strain
The team crafted hollow, curved MoSSe nanoshells (a mixed MoS₂/MoSe₂ “MSS”) that are biaxially strained up to ~1.8%. Curvature + strain do three things:
- Expose low-coordination Mo sites on the basal plane (normally hidden by chalcogen layers).
- Create S/Se vacancies more easily, which increases surface activity and conductivity.
- Shift Mo d-band and chalcogen p-band centers upward, strengthening d–p hybridization with sulfur (S*–Mo bonds) and s–p interactions with Li—both of which accelerate charge transfer.
Translation: the surface gets more “sticky” and more “electrically chatty,” so polysulfides latch faster and convert quicker.
What changed in practice
- Adsorption mode flips from weak Li–S/Se to strong S–Mo bonding* on strained MSS.
- Lower activation barriers across key voltages (notably near ~2.0 V where Li₂S nucleation is rate-limiting).
- Reduced polysulfide shuttle constant and longer, flatter plateaus at higher current—signs of better sulfur utilization.
- Device-level wins: a multilayer pouch cell with ~6.11 Ah initial capacity and ~396 Wh/kg (all components weighed) at E/S ~3.5 µL mg⁻¹; coin cells held >600 mAh g⁻¹ at high rates with ~3× lower fade than controls.
For Non-Scientists: a quick analogy
Picture a racetrack (the battery). The race cars (sulfur species) keep drifting into the pits (the wrong side of the battery), wasting fuel. Engineers reshaped the track surface so hidden “magnets” (Mo sites) poke through. These magnets grab the cars just enough to guide them through the turns faster and keep them on the right side of the track. The race finishes quicker, wastes less fuel, and the cars hold up longer.
Why this matters now
If Li–S moves from lab cells to large pouch cells with lean electrolyte and repeatable cycling, we edge closer to lighter packs for drones, aviation, and EVs, and lower-cost storage (sulfur is abundant). This paper shows an atom-level design rule—use biaxial strain as a single geometric descriptor to predict and enhance sulfur redox kinetics—so others can replicate and iterate.
What to watch next
- Scale & stability: Can manufacturers grow these strained nanoshells uniformly at kilogram scale?
- Anode safety: Pairing with stabilized lithium metal (coatings, hosts, or solid electrolytes).
- System energy density: Crossing >400–500 Wh/kg at the pack level with >200 cycles in real duty profiles.
Check out the cool NewsWade YouTube video about this article!
Article derived from: Yang, JL., Xu, H., Xiao, T. et al. High-dimensional strain unlocks fast polysulfide redox kinetics for lithium-sulfur batteries. Nat Commun 16, 8910 (2025). https://doi.org/10.1038/s41467-025-63969-z