Sodium-Ion Batteries Just Got a Major Upgrade—Thanks to a Microscopic Tweak
Lithium-ion batteries have dominated the market for years, but sodium-ion batteries are rising fast as a cheaper, more sustainable alternative. However, they face one critical challenge—layer instability caused by a phenomenon known as “inter-layer gliding.” Now, scientists may have cracked the code to solving this problem using a technique called interphase engineering.
A new study published in Nature Communications unveils a groundbreaking strategy to suppress this gliding behavior and extend the life and performance of sodium-ion batteries. Let’s dive into how this innovation works—and why it matters.
What’s the Problem with Layered Oxides?
At the heart of sodium-ion batteries are materials known as layered transition metal oxides. These are structures made of stacked metal oxide sheets with sodium ions shuttling between them during charging and discharging.
But there’s a catch: during this cycling, the layers tend to shift or glide, leading to unwanted phase transitions—a change in the material’s internal arrangement that can degrade the battery’s performance over time.
The Breakthrough: Interphase Engineering
To combat this issue, researchers introduced a clever solution: interphase engineering.
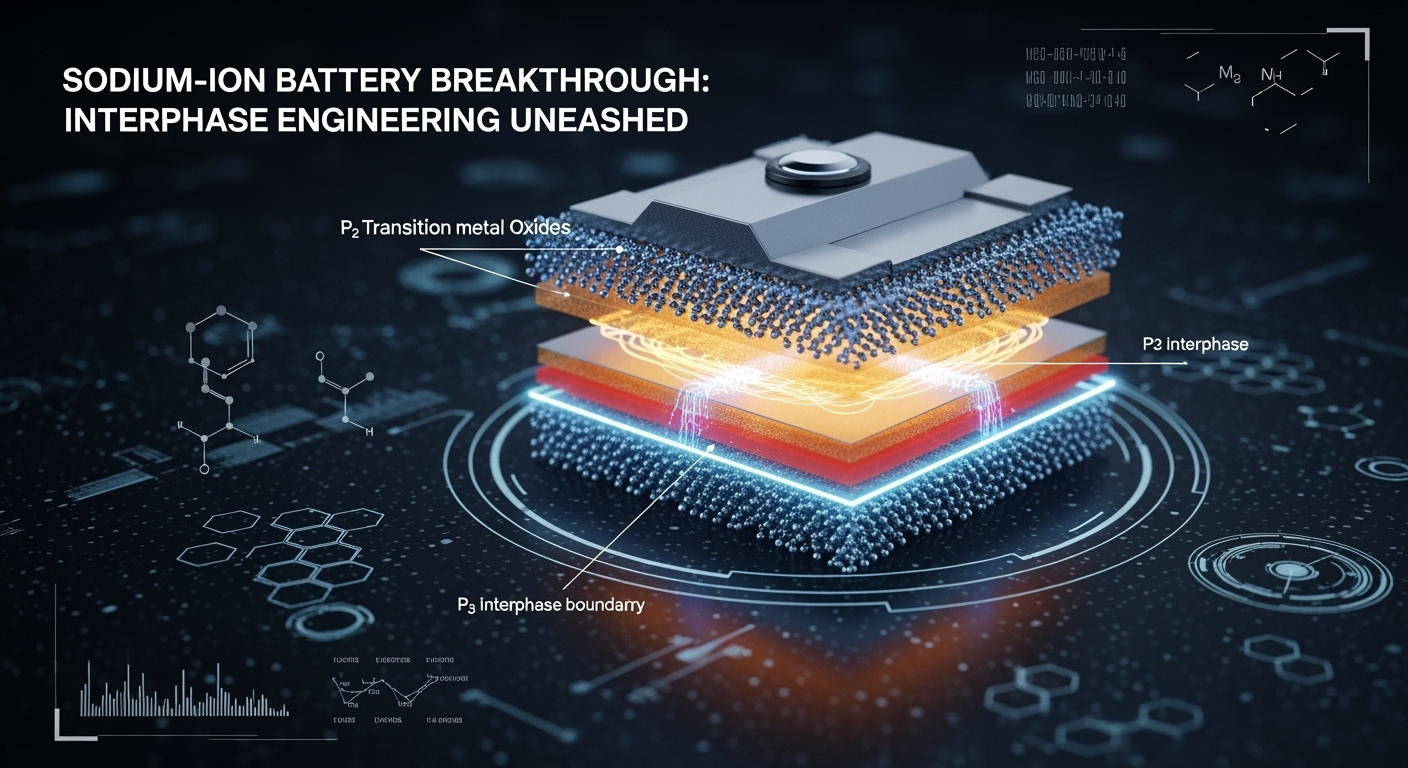
They created a new material called P2/P3-Na₀.₄₆Mn₀.₉Ni₀.₁O₂, which strategically combines two types of crystal phases—P2 and P3. Where these phases meet, they form what scientists call a X2/Y3 interphase. This boundary behaves like an internal anchor, preventing the layers from gliding out of alignment.
Even better, this interphase remains energetically stable throughout charging and discharging, meaning it resists structural change and keeps everything locked in place.
Why This Is a Big Deal
The team’s engineered material showed:
- 81.2% capacity retention after 500 cycles (compared to 67%–69% for non-engineered materials)
- Improved rate capability, meaning faster charging without performance loss
- Better structural stability, confirmed by atomic-level microscopy and synchrotron X-ray diffraction
In other words, this tiny structural tweak results in big performance gains.
The Science Behind the Stability
The key lies in the fact that P2 and P3 phases can’t easily transform into each other during operation, which locks their shared interphase in place. This immobile boundary prevents the gliding of MO₂ layers—an issue that causes structural fatigue in other battery materials.
To quantify and control this effect, the researchers introduced two new metrics:
- Interphase density (α): How often these stabilizing boundaries appear in the material.
- Interphase uniformity (β): How evenly these boundaries are distributed.
A high α and low β means better suppression of phase transitions and better battery performance.
A New Direction for Battery Design
The implications of this study go beyond just sodium-ion batteries. It suggests a new design principle for high-performance energy materials: rather than simply mixing different phases together, engineers can deliberately construct interphases to suppress structural degradation.
It’s not just about what goes into the battery—but how those ingredients are arranged at the atomic scale.
What’s Next?
The researchers tested their approach in multiple material combinations and consistently saw improved cycling performance. However, perfecting the distribution of these interphases (maximizing α and minimizing β) remains a challenge.
Still, this discovery offers a powerful roadmap for designing next-gen batteries that are:
- Longer-lasting
- More affordable (thanks to sodium)
- Environmentally friendlier than lithium-based options
Final Thoughts
As the world races toward a future powered by clean energy, the need for better, safer, and cheaper batteries is more urgent than ever. Interphase engineering could be the secret weapon to unlock the full potential of sodium-ion batteries—and reshape the way we think about energy storage.
Check out the cool NewsWade YouTube video about this article!
Article derived from: Zhou, X., Yang, C., Liu, X. et al. Inhibiting inter-layer gliding in transition metal layered oxides through interphase engineering for sodium-ion batteries. Nat Commun 16, 6691 (2025). https://doi.org/10.1038/s41467-025-61065-w